User:Mr. Ibrahem/Floxuridine
 | |
| Clinical data | |
|---|---|
| Synonyms | 5-fluorodeoxyuridine |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682006 |
| Pregnancy category | |
| Routes of administration | Intra-arterial |
| Drug class | Antimetabolite[1] |
| Identifiers | |
| |
| Chemical and physical data | |
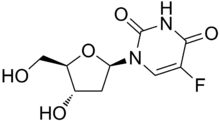
| Formula | C9H11FN2O5 |
| Molar mass | 246.19 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 150.5 °C (302.9 °F) |
| |
| |
| (verify) | |
Floxuridine is a medication used to treat liver cancer and gastrointestinal adenocarcinoma or colorectal cancer that has spread to the liver.[1][2] It is given by gradual injection into an artery supplying the cancer.[1]
Common side effects include bone marrow suppression, tiredness, headache, dizziness, trouble sleeping, numbness, abdominal pain, constipation, diarrhea, heart burn, nausea, mouth inflammation, and rash.[2] Other side effects may include liver problems.[2] Due to the severity of side effects people are generally admitted to hospital during initial treatment.[1] It is an antimetabolite, specifically a pyrimidine analog.[1]
Floxuridine was approved for medical use in the United States in 1970.[1] It is available as a generic medication.[2] In the United States 500 mg did costs about 130 USD; though the only manufacturer stopped making the medication in 2019.[3][4]
References
[change | change source]- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 "Floxuridine Monograph for Professionals". Drugs.com. Archived from the original on 16 August 2019. Retrieved 11 December 2021.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 "Floxuridine". LiverTox: Clinical and Research Information on Drug-Induced Liver Injury. National Institute of Diabetes and Digestive and Kidney Diseases. 2012. Archived from the original on 11 December 2021. Retrieved 11 December 2021.
- ↑ "Floxuridine Prices, Coupons & Patient Assistance Programs". Drugs.com. Archived from the original on 17 April 2021. Retrieved 11 December 2021.
- ↑ "Drug Shortage Detail: Floxuridine Injection". www.ashp.org. Archived from the original on 18 September 2021. Retrieved 11 December 2021.
